What makes you smile ?
Facial reanimation
Facial paralysis is a highly burdening situation, which is associated with emotional distress and overall reduced quality of life. In patients who still suffer from paralysis after a period of observation or following medication, surgical methods are taken into consideration. Our team proposes a new, less invasive approach for dynamic facial reanimation post facial paralysis using soft artificial actuators. The proposed solution relies on the use of DEAs instead of biological muscles to restore mouth and more precisely smiling movement.

Artificial Bio-Bladder

The human bladder is an expandable muscular organ which stores urine and help in its flow control. Various diseases can occur that could prevent the bladder from a proper function. To develop new treatments, a better understanding and appropriate methods are needed. . Therefore, an in-vitro artificial bladder based on dielectric elastomer actuators is under development which mimics the function of the bladder to prevent cell cultures from dedifferentiation.
© Werner Siemens Stiftung, Félix Wey
Artificial urinary sphincter
Urinary incontinence is a prevalent medical condition that affects millions of individuals worldwide, transcending age, gender, and demographic boundaries.
Among the various treatment available, the development of an Artificial Urinary Sphincter (AUS) represents a significant progress towards improving the treatment of urinary incontinence. Within the Center for Artificial Muscle (CAM), we are developing an AUS based on a Dielectric Elastomer Actuator (DEA).

Valveless Impedance-driven DEA Pump for Fontan failure Support
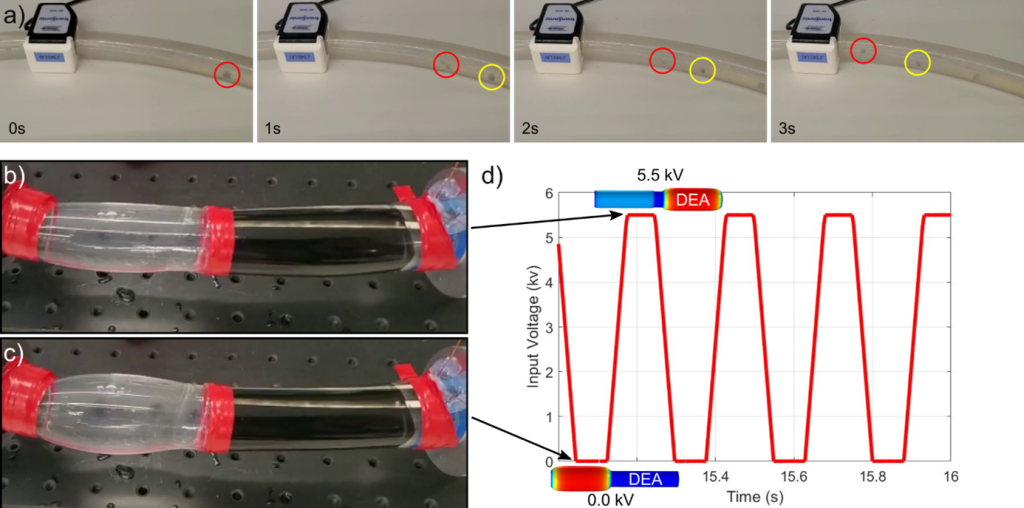
We aim to investigates the feasibility of an innovative valveless impedance-driven pumping device based on a fully integrated actuation (DEA). The primary focus of this device’s application is aimed at addressing Fontan failure, a condition where the conventional passive conduit used in the Fontan procedure could potentially be replaced by an active conduit, which in this case is the valveless DEA pump. This replacement offers a promising solution to alleviate the complications associated with Fontan failure. The pump itself is composed of an active DEA tube connected to a passive tube through a decoupling link made of PMMA.
First -in vivo- augmented aorta
Scientists from EPFL and University of Bern have successfully implanted – in vivo – their first artificial tubular muscle that augments the aorta and assists cardiac function in pumping blood.
© Werner Siemens Stiftung, Félix Wey
Dielectric Elastomer Acuator Manufacturing

We have established a new process flow to manufacture multilayer actuators starting from the commercial Elastosil film (100 or 200um thick) from Wacker Chemie. We obtained an actuator composed of at least four layers. Thanks to a neat trick (Patent pending CH20190012), we are able to reach the electrical limit of the commercial material for a multilayer actuator.
DEAs test and modelling
We have developed an automatic test bench to evaluate and perform intensive tests of our actuator. The preliminary assessments show promising features both in terms of the actuator characteristics and the test bench capability. We have compared these measurements to our Finite Element Model to validate the latter. We now have a near-predictive model that will be useful to optimize the final actuator.
© Werner Siemens Stiftung, Félix Wey

Flowloop and Windkessel model

We have a mock-up Flow loop that imitates the blood circulatory system. We have undertaken our first attempt to plug our actuator on the flowloop.
In parallel, we have established a more accurate, lumped model that is based on the Windkessel model and in which we have integrated our actuator model. This tool will allow us to forecast the blood circulatory system’s response according to our actuator activation. The preliminary measurements on the flowloop will allow us to adjust our modified lumped model to match the body’s behavior as closely as possible.
The lumped model has also highlighted that one ring might be a good starting device to support the heart. It allows us to modify the pressure after the aortic valve. We have been able to demonstrate that we are capable of modifying the heart’s energy or blood flow rate by approximately 10 % depending on the way we actuate our device.
© Werner Siemens Stiftung, Félix Wey
Power supply
We already have a custom-made, compact set-up showing the wireless power transfer working on a battery. We have also developed our own electronic system going from 12 V to 7 kV. This electronic should also allow us to harvest the electrical energy stored in our actuator in order to increase the global efficiency of our system. We have just started to plug the new electronic to our actuator.
We plan to merge the two electronic subsystems in 2020 resulting in an efficient power supply.
© Werner Siemens Stiftung, Félix Wey

